A Passion Avenue For Science
Recent surge in pharmaceutical micro-pollutants in water bodies calls for an efficient method to neutralize wastewater to sustain the ecosystem. One of the ways to degrade drug molecules is through photocatalytic degradation using UV rays. ZnO is known to be a common catalyst in the degradation of contaminants found in wastewater. However due to its toxicity to the environment, there is a need to objectively re-evaluate its necessity and alternatives.
Although the current wastewater treatment plant (WWTP) can neutralize up to approximately 95% of these pharmaceutical components, pharmaceutical micro-pollutants still end up in the environment at low concentrations, in which its toxicological effects remain.
Information
Indonesia's Hospital Wastewater: 36% is treated, 64% is dicharged directly to water bodies without treatment.
Ibuprofen: the world's 3rd most consumed drug, a dominating pharmaceutical micro-pollutant and highly toxic for the environment.
Clotrimazole: highly toxic for aquatic ecosystem; concetrations of 20ug/L can cause lethal effects on crustaceans.
Conclusion and Recommendations
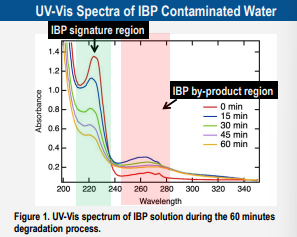
Although it was generally perceived that the addition of ZnO as photocatalyst increases the effectiveness of UV treatments for degrading pharmaceutical micro-pollutants, our work reveals that there are various outcomes that require a careful look. We found that IBP was not as efficiently degraded when ZnO was used in the solution during the UVC treatment with maximum degradation potential of 86.6% compared to that without ZnO at 94.4%. Despite the ability of ZnO to degrade the IBP faster, this advantage is superseded by its inability to degrade the IBP by-product. In other case such as in CTZ, high degradation efficiency with and without ZnO are observed. The use of ZnO for degrading CTZ appears to be better than that without with maximum degradation potential of 99.9% and 92%, respectively. Optimal ZnO concentration was determined to be ~ 1 g/L, above which, screening effects due to an increased turbidity starts to dominate the system which lowers the photodegradation effective-ness. Overall, it is clear that UVC without ZnO is sufficiently effective in degrading IBP and CTZ with a maximum degradation potential of more than 90% in both cases. Furthermore, the by-product of IBP is also shown to be more effectively degraded without ZnO. Thus, we hope that our results may further encourage the adoption of a simple UVC batch-stirred treatment system to treat wastewater in households, hospitals, and pharmaceutical industries.
In this work, Shania determined to compare the effect of ZnO catalyst in degrading several drug wastes in waters.
Comparative Analysis of Photodegradation of Ibuprofen and Clotrimazole Water Pollutant using UVC Rays in Presence and Absence of ZnO Photocatalyst
2022